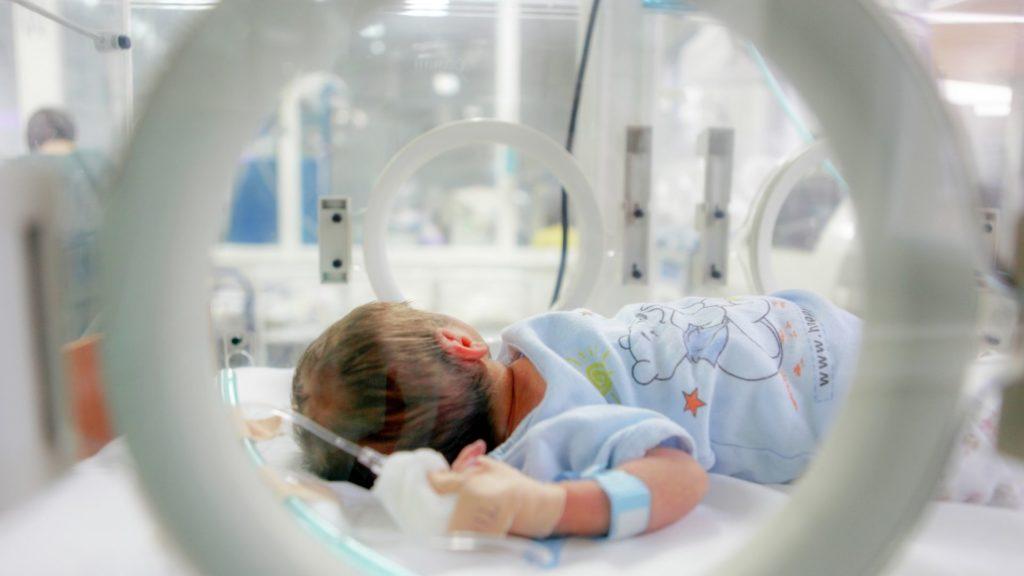
Welcome to 99nicu – Your Dedicated Community for Neonatal Care Professionals
At 99nicu, we offer more than just quick exchanges. We provide a space for lasting discussions for neonatal health care professionals. As a member, you’ll join a global network of NICU staff focused on exchanging clinical experiences and best practices.
Unlike fast-paced social media platforms, our community is designed for meaningful, sustained conversations. Find organized forums, webinars and valuable resources that you can revisit and search anytime. Our focus is long-term engagement, ensuring that each interaction contributes to your professional development and the advancement of neonatal care in your context.
Join today and experience a space where your expertise is valued and your contributions make a lasting impact.
Featured Content
Hot Forum Topics
Community News
Upcoming Events
-
Course in neonatal lung ultrasound (in French)
Le NeoGEPP annonce une nouvelle formation!!
L'échographie pulmonaire est devenue un outil essentiel dans la prise en charge du nouveau-né malade. Rejoignez-nous pour notre prochaine formation au CH Versailles le 16/06/2026 avec focus sur l'échographie pulmonaire néonatale et une introduction à l'échographie hémodynamique.
Sessions pratiques sur simulateur et au lit du malade.
Inscriptions et informations sur 👉👉www.groupegepp.fr (Rubrique formation)
🗓️ Le 16/6/2026
📍CH A. Mignot à Versailles (France)
💶 250 euros (déjeuner compris)
🇫🇷 En français
Places limitées!
Contact neogeppfrance@gmail.com
et sur le site www.groupegepp.fr
Programme 160626 NeoGEPP POCUS Versailles 2026 .pdf
- 0 comments
-
Perinatal Care of the Preterm Baby Epidemiology and Ethics
Perinatal Care of the Preterm Baby-Epidemiology and Ethics
This is an online module being organised by the MPROvE Academy starting from the 12th of February till the end of April 2021. The content covered includes limits of viability, prenatal counselling, communication, prognostication, decision making, and a lot more as outlined below. The course has been broken up into content that can be imbibed weekly with a webinar covering that topic. The course has online content, and videos for review by the participants. Participants can access this from anywhere in the world. For more details a video of the course is attached.
For registration please contact Dr Alok Sharma Consultant Neonatologist on draloksharma74@gmail.com
- 0 comments
-
Certification in Neonatal Therapy 2021
Certification in Neonatal Therapy 2021
Requirements for Certification in Neonatal Therapy include Qualified and Licensed Therapist who is holding Elite membership of Association of Neonatal Therapists. He/she should have completed 40 hours of training in neonatal therapy/care (direct teaching and or webinar), upto 350 hours of Direct Mentoring in Affiliated Set up. Kindly use following checklist for your reference.
1) Completed Post Graduation (Final year student can apply)
2) Active License
3) Elite Member of Association of Neonatal Therapists
4) Active professional membership of ANT
5) 40 hours of training in neonatal therapy (direct teaching / online webinar) ...scrutiny about validity of the courses will be done.
6 ) 350 + hours of Direct mentoring in affiliated set up - after examination
Cost Rs. 40000/- (Approximately)
7) Exam fees INR 1000/- Rs
😎 TRAIN Workshop completion (during mentorship)
- 0 comments
-
IPOKRaTES Clinical Seminar: Best care for tiny babies
Objectives
This seminar is to share knowledge, to understand the latest advances and to develop collaborations and networks to provide the best care for tiny babies, their families and their communities.
For further information, please visit the website www.ipokrates.info
- 0 comments
Professional Development
Latest Research From Neonatal Journals
Latest in Links Directory
Who's Online (See full list)
Member Statistics
- 7,214 Total Members
- 2,455 Most Online
-
Vladislav Raclavsky Newest Member ·
_0b6494.png)
_5c07e1.png)
_0d1bd3.png)
_75149d.png)









.png.cbe08663ecd07901cfa3df279a1e4d5b.png)















.jpg.59ab5a1ef4cde9744d175a335a798aac.thumb.jpg.f1c20d41d563c7b7a5dad4d4cfb7848e.jpg)