-
Vancomycin therapeutic drug monitoring in the NICU - methods, targets, thoughts?
Hi all, This is an open request to the community to share your policy about vancomycin therapeutic drug monitoring in your units. I anticipate a lot of variability 🙂 You may follow my structure or write your take freeform. Indications:Suspected or proven infection with gram-positive bacteria, usually MRSA. Also as prophylaxis around surgery (often as a single dose). Dosage:We currently strive to administer a loading dose of 20 mg/kg (this is a relatively new practice for us, for years our neonatologists refrained from vancomycin loading doses, possibly due to habit/tradition) regardless of infant age and renal function to reach steady state as quickly as possible. According to Jacqz-Aigrain et al., J Antimicrob Chemother, 2019, a loading dose of 25 mg/kg significantly increased the percentage of neonates reaching the target AUCss–24 of 400 mg·h/L. We prefer the slightly lower dose of 20 mg/kg. Following the loading dose we would administer doses of 15 mg/kg if no renal impairment is apparent, and we use NeoVanco to select the most appropriate dosing interval. NeoVanco is a freely available prediction tool developed by Adam Frymoyer, MD (Stanford University - Stanford Children's Health) according to a population pharmacokinetic model developed by Frymoyer A et al. (2014). It is not a 'full' Bayesian calculator, but rather a prediction tool for starting treatment (it is not possible to enter vancomycin serum levels to tailor dosage to the individual infant). It provides the estimated AUC and Ctrough for several dosage regimens, as well as the probability to reach AUC > 400 mg·h/L and Ctrough > 20 mg/L at steady state. Monitoring:In infants with no apparent renal impairment, we draw serum levels before the 4th dose. In extremely preterm infants (< 28 GA) or infants with renal impairment, we would draw serum levels before the 2nd dose. The most recent guidelines on vancomycin dosing and monitoring are the Rybak et al. guidelines (2020), which provide recommendations for adults, pediatric and neonatal patients, although most of the neonatal recommendations are based on the pediatric recommendations and it is stated that data are limited in that population. Dosing adjustment:Based on serum levels we may adjust the treatment regimen, preferably by adjusting dosing interval and not the dose (to avoid high peaks and troughs), although that may not always be possible/desired (sometimes we would prefer longer intervals to minimize tampering with the IV line to reduce risk of infections). We find the dosing adjustment recommendations of the Australasian Neonatal Medicines Formulary (ANMF) convenient and sensible. Limitations:We do not use a dedicated Bayesian vancomycin calculator (many Bayesian calculators we are aware of require a paid subscription and most do not offer a dedicated PK model for neonates. Additionally, our volume of vancomycin treatments may not justify such an expense), so in essence we still rely on Ctrough, which is not recommended anymore. We hope that the combination with the NeoVanco predictions helps us to avoid toxicity. We tried to utilize 1st order PK equations as an alternative to Bayesian calculations, but these require timed measurements and both peak and trough measurements, which complicate monitoring too much. We do not implement continuous infusions of vancomycin, even though in certain clinical scenarios it may seem suitable. This is still under debate in the literature. Speaking of literature, there are so many papers and texts of vancomycin monitoring in general and in neonates, that it is so easy to get lost and confused about the optimal course of action. Please share your thoughts and methods for the benefit of the community, thanks!
-
"Reducing IVH in Preterm Infants Through Optimized Inotropic Support" - a Webinar by the Newborn Brain Society (NBS), Thursday, 28.5.26, 12pm EST
Join the Newborn Brain Society for this week's QI Educational Webinar session with guest speaker, Sujith Gurram, on Thursday (5/28) at 12pm EST: "Reducing IVH in Preterm Infants Through Optimized Inotropic Support" Registration is FREE, but please RSVP below to confirm your attendance and receive the link to attend: https://us02web.zoom.us/webinar/register/WN_TRxZzDcvRKO5VIrzmmwszw#/registration For more NBS webinars, visit their website: https://newbornbrainsociety.org/webinars-and-sessions/
-
Dotan S changed their profile photo
-
A webinar by the Irish Medicines in Pregnancy Service (IMPS) - "Supporting Safe and Effective Medicines Use in Pregnancy and Breastfeeding - Balancing Benefit and Risk"
The Irish Medicines in Pregnancy Service (IMPS) at Rotunda Hospital, Dublin, hosted a webinar last week titled: "Supporting Safe and Effective Medicines Use in Pregnancy and Breastfeeding - Balancing Benefit and Risk". The recording and slides are currently available here. The webinar dealt with risk-benefit decision making in pregnancy, evaluating information, medication use in breastfeeding and risk communication and shared decision making. In addition, the speakers shared several sources for drug information in pregnancy and breastfeeding. All material is freely available, highly recommended to watch.
-
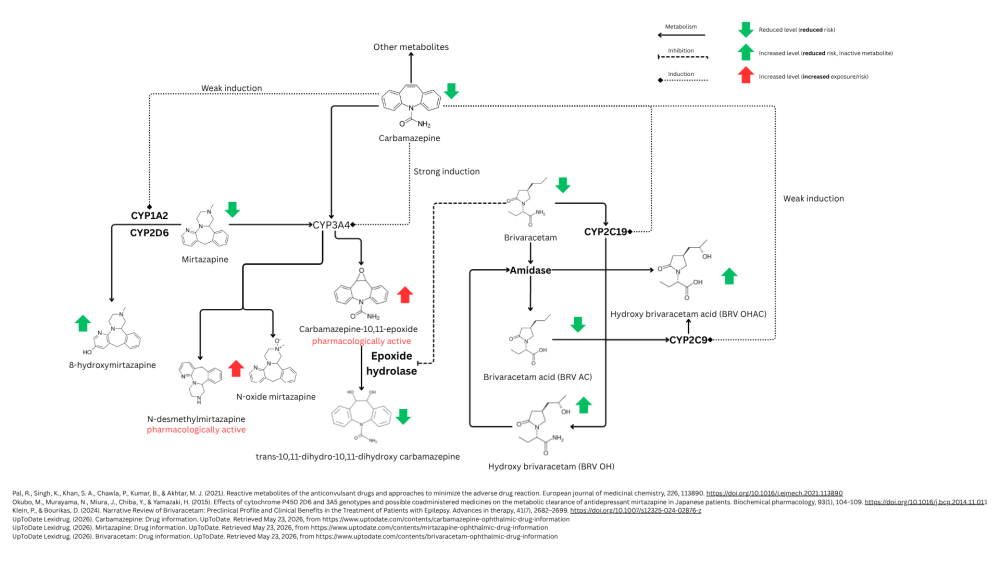
Feeding practice in mother with polypharmacy.
Hi Gustaf, This is indeed a complex clinical scenario that should be approached from several angles - mother, infant, drug and feeding. This approach was discussed only a few days ago in a webinar hosted by the Irish Medicines in Pregnancy Service (IMPS) (available for watching here, highly recommended). Taking these four pillars into consideration will help optimize care for both mother and infant. First, the drugs in question - let's summarize what we know about the metabolism of these drugs: • Carbamazepine is metabolized in the liver, mainly by the CYP3A4 enzyme, yielding the pharmacologically active metabolite carbamazepine-10,11-epoxide (as well as several other non-active metabolites). Carbamazepine also induces the activity of CYP3A4, CYP1A2 and CYP2C19 to varying degrees (primarily CYP3A4). • Brivaracetam is primarily metabolized by a CYP-independent hydrolysis and secondarily by hydroxylation mediated by CYP2C19. It also inhibits the enzyme epoxide hydrolase, which hydrolyzes the carbamazepine-epoxide metabolite. • Mirtazapine is metabolized via several hepatic enzymes: CYP2D6, CYP1A2 and CYP3A4. One of its metabolites is known to be pharmacologically active (N-desmethylmirtazapine). ** Please see the attached diagram for better clarity (hopefully...) ** Assuming steady state for all drugs, from this metabolism overview we can appreciate that maternal levels of brivaracetam and mirtazapine are expected to be lower than reported in the absence of carbamazepine, which is reassuring for mirtazapine, whose milk levels were not predicted to be high to begin with. We can put it aside and not worry about it. For brivaracetam, data on passage into breastmilk is limited, and the dose is very high (even supra-therapeutic per common guidelines), so milk levels would also be expected to be rather high, even if the overall AUC of the drug is somewhat decreased due to enzyme induction by carbamazepine. So as an interim summary: from a PK point of view, we are not worried about mirtazapine or its pharmacologically active metabolite N-desmethylmirtazapine. While the levels of the latter are expected to be higher in this scenario due to induction of CYP3A4, its baseline passage into milk is still expected to be rather low (cf. Kristensen et al. (2007). Transfer of the antidepressant mirtazapine into breast milk. British journal of clinical pharmacology, 63(3), 322–327. https://doi.org/10.1111/j.1365-2125.2006.02773.x). However, we are not comfortable with brivaracetam because its levels may still be relatively high. Now for carbamazepine: The parent drug is considered compatible with breastfeeding, although both it and its active metabolite (carbamazepine-10,11-epoxide) are known to pass into breastmilk (cf. Kacirova et al. (2022). Carbamazepine and carbamazepine-epoxide concentrations in mothers, colostrum, and breastfed newborns: Comparison with concentrations determined during delivery and in the mature milk period. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie, 151, 113176. https://doi.org/10.1016/j.biopha.2022.113176). Considering the fact that the maternal dosage is rather high, this may translate into relatively high milk levels for both. Lactmed mentions that sedation, poor sucking, withdrawal reactions and 3 cases of hepatic dysfunction have been reported following exposure to carbamazepine in breastmilk. This is complicated by the fact that brivaracetam inhibits epoxide hydrolase, effectively increasing the risk for higher levels of carbamazepine's active metabolite, which has anticonvulsant and neurotoxic effects. So we could anticipate a higher exposure to the active metabolite. If we try to summarize pharmacokinetic interactions data, we may expect relatively high exposure to carbamazepine and brivaracetam based on high maternal doses, and to carbamazepine epoxide based on inhibition of its metabolism, alongside some unpredictable exposure to the active N-desmethylmirtazapine as mentioned above. All of these exposures may translate into adverse CNS effects in the infant, i.e., lethargy, poor sucking, impaired weight gain and such. Additionally, neonatal hepatic function should be taken into account, which MIGHT be lower than normal due to prematurity - most CYPs function at a fraction of adult function (usually 10-30% in the first weeks of life, depending on gestational age and comorbidities, and then slowly increasing with overall development). We can assume that neonatal clearance may be somewhat lower than a term infant, which may contribute to the already high exposure we anticipated. On the other hand, infant and maternal characteristics should be taken into consideration: This is a premature infant that needs human milk for optimal overall development, normal development of the immune system and lowering risk for NEC. In addition, he is in the NICU, under constant monitoring. Furthermore, the mother's medical condition is controlled and she is willing to breastfeed. Under proper guidance, this would benefit both mother and infant, not only physically, but also emotionally and would consolidate bonding and wellbeing for both. Therefore, the benefits of human milk provision in this case may still outweigh the risks mentioned above, but it should be done prudently. After integrating drugs, infant and maternal factors, I would suggest implementing mixed feeding, i.e., alternating between mother's own milk and donor milk (if available) at a ratio that seems reasonable for all involved ("start low, go slow"). @Mariana Oliveira rightly pointed out that you have already allowed him to be exposed to mother's own milk in small amounts with no adverse effects, so this is reassuring, but when talking about higher volumes, careful exposure management is still recommended. This may probably be the best course of action here until the infant's response to such potentially higher exposure would be more apparent. At any rate, mother should also avoid night feedings, as an epileptic postpartum patient must have regular, uninterrupted sleep to prevent seizures and to be able to function during the day. So the non-exclusive feeding pattern is essentially already there, and alternating between mother’s own milk and donor milk during the day may be optimal, at least until things get clearer. Then - if the infant shows no signs of adverse effects that may be attributed to the drugs - you could consider gradually increasing the volume of human milk over donor milk, hopefully up to exclusive or almost exclusive breastfeeding. I would refer here to @Mariana Oliveira's suggestion to time feeding in accordance with drugs PK characteristics (half-life, Tmax, etc.). This is indeed a valid approach, which can aid in reducing exposure, e.g., not feeding within Tmax, which is the period when drug levels are building in serum and milk, or attempting to time feedings when drugs are at minimal levels (trough). The only caveat here is that these methods work best when feeding schedule is more flexible and controllable, i.e., in older infants. In the NICU, when infants feed every 2-3 hours, it may be difficult to time feedings in accordance with drug characteristics. But this is definitely worth considering, and even if it is not feasible now, it may be feasible in the future when the infant grows (also if something changes, i.e., a new drug is introduced). The infant should be monitored as is customary in the NICU, also by the mother, who should learn to identify his habits, sleeping patterns, etc. Due to anticipated high exposure to carbamazepine, infant’s liver enzymes should be monitored regularly. Therapeutic drug monitoring for carbamazepine (which I assume is available in your facility) should also be considered for the infant, but this may be reserved for use only if other markers suggest high exposure, to avoid excessive blood draws, pain, etc. Finally, while the wellbeing and seizure control for the mother are of the utmost importance, seeing how she is controlled and seizure-free, I agree with @Mariana Oliveira that you could consider involving her neurologist to discuss lowering her dosage(s) carefully, to reduce infant exposure through milk while keeping the mother well. This is delicate of course, but might be considered via a joint discourse with her doctors (primary physician, neurologist) AND the mother herself. Please keep us posted, if possible. It would be interesting to know what transpires. If you happen to measure carbamazepine serum levels for mother and infant, we could even calculate relative infant dose (RID). Theoretically, this can also be estimated using published serum levels in, e.g., Lactmed, although serum levels under such doses as in this scenario are not available. Moreover, their warning regarding anticonvulsant medications should be heeded: "In published reports of anticonvulsant use during breastfeeding, most women were taking a combination of anticonvulsants. Some other anticonvulsants (e.g., phenytoin, carbamazepine) stimulate the metabolism of other drugs including anticonvulsants, whereas others (e.g., valproic acid) inhibit the metabolism of other drugs. Therefore, the relationship of the maternal dosage to the concentration in breastmilk can be quite variable, making calculation of the weight-adjusted percentage of maternal dosage less meaningful than for other drugs in this database." [Lactmed - Brivaracetam]. Good luck!
-
Polyethylene glycol (macrogol) use in extremely preterm infants
We do not use it. What is the indication?
- Hydrocortisone for infants < 23 weeks
-
Preparation of drugs in standard concentrations in the NICU
We have published our recommended standard concentrations in a position statement last year. While the majority of the statement is in Hebrew, the recommendations are in English and are attached below. The full text can be found here and can be translated using AI/Google Translate as needed (or you can ask me for any clarifications). I also played a bit with Gemini recently and created this "NICU Smart Infusion Tool" (also attached, should work offline from any browser, just download and open in browser - free to use and share). This tool takes as input a medication from a predetermined list of drugs administered continuously in the NICU, infant weight and dosage OR delivery rate, and calculates the pump rate, delivered dose and total volume of fluid per day as output for each of the standard concentrations. These calculations complement the recommendation for using standard concentrations and should help the clinician choose the most appropriate concentration for any individual patient. It should be noted that the various concentrations were originally meant for different weight categories, but with accumulated experience we have found that for e.g. Fentanyl, using the 2.5 mcg/mL for very small babies may actually load them with excess fluids, so in practice we use 5 or 10 mcg/mL (using the tool as a decision supporting system). So while all of the recommendations are evidence-based, some are pending revision, but can still be used at the discretion of the clinician. For other common drugs administered as intermittent infusion, we use the following concentrations: Ampicillin: 25 mg/mL or 50 mg/mL (when higher doses are needed, e.g., for meningitis, or for very large babies). Caffeine: 5 mg/mL or 10 mg/mL (when a loading dose is required) Fluconazole: 2 mg/mL or 1 mg/mL (for very small babies) Darbepoetin: 40 mcg/mL And in general, even for rarely administered drugs with no established standard concentration, we try to prepare them in a concentration that will be easy to calculate, easy to dilute down to and produce a volume that will be easy to administer (i.e., not too small). NICU Smart Infusion Tool.html
-
Fluconazole prophylaxis
Our guidelines (a mid-size hospital in Israel with a 25-bed NICU) also instruct to give 3 mg/kg every 72 hours for eligible infants and decision to treat is based on the presence of a central line of any kind as a primary determinant and on corrected age/birth weight as a secondary determinant. There are other secondary determinants if the above criteria are negative (NEC diagnosis, prolonged treatment with broad-spectrum antibiotics), see the attached diagram. Here are our references: Rolnitsky, A., Levy, I., Sirota, L., Shalit, I., & Klinger, G. (2012). Targeted fluconazole prophylaxis for high-risk very low birth weight infants. European journal of pediatrics, 171(10), 1481–1487. https://doi.org/10.1007/s00431-012-1760-2 Pappas, P. G., Kauffman, C. A., Andes, D. R., Clancy, C. J., Marr, K. A., Ostrosky-Zeichner, L., Reboli, A. C., Schuster, M. G., Vazquez, J. A., Walsh, T. J., Zaoutis, T. E., & Sobel, J. D. (2016). Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America, 62(4), e1–e50. https://doi.org/10.1093/cid/civ933 Ericson, J. E. et al. (2016). Fluconazole Prophylaxis for the Prevention of Candidiasis in Premature Infants: A Meta-analysis Using Patient-level Data. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America, 63(5), 604–610. https://doi.org/10.1093/cid/ciw363 Rios, J., Camargos, P., Corrêa, L. P., & Romanelli, R. (2017). Fluconazole prophylaxis in preterm infants: a systematic review. The Brazilian journal of infectious diseases : an official publication of the Brazilian Society of Infectious Diseases, 21(3), 333–338. https://doi.org/10.1016/j.bjid.2017.01.008 Ericson, J. E., & Benjamin, D. K., Jr (2014). Fluconazole prophylaxis for prevention of invasive candidiasis in infants. Current opinion in pediatrics, 26(2), 151–156. https://doi.org/10.1097/MOP.0000000000000060 Zhang, D., Xie, D., He, N., Wang, X., Dong, W., & Lei, X. (2021). Prophylactic Use of Fluconazole in Very Premature Infants. Frontiers in pediatrics, 9, 726769. https://doi.org/10.3389/fped.2021.726769 Fanaroff & Martin's, Neonatal-Perinatal Medicine, Diseases of the Fetus and Infant, 11th Edition Pages 825-827
-
Hydrocortisone for infants < 23 weeks
Hi all, Yesterday, the Tiny Baby Collaborative held a webinar about using hydrocortisone in extremely preterm infants (under 23 weeks of gestational age), which featured Dr. Satoshi Kusuda from the Neonatal Research Network of Japan and Dr. Erik Jensen from Stanford University (the webinar will probably be available in the near future here: https://www.tinybabycollaborative.org/webinars) I was unable to attend the entire event, but was wondering if anyone here also attended and maybe we can discuss this issue. My understanding is that in such extremely preterm infants, adrenal insufficiency is very common (which makes sense) and with the added stress of premature delivery and adaptation, circulatory collapse is a common risk. The two experts presented several papers and reviews, but I am afraid I am not clear on the bottom line (beside the fact that we need more studies, as always). Hydrocortisone was shown to reduce BPD and failure to extubate, as well as in-hospital death, but was shown to increase the risk for hyperglycemia. A comparison with dexamethasone was also shown, where dexamethasone was shown to have a lower risk ratio for failure to extubate (0.61 vs. 0.79 for hydrocortisone). The experts concluded that no robust data inform use of hydrocortisone for respiratory or neurodevelopmental indications in infants born before 23 weeks of gestation. Interested to hear your thoughts about this, as well as your practices. How do YOU use hydrocortisone (or other corticosteroids) in tiny infants? Here are some of the papers that were cited in the webinar: Effect of Prophylaxis for Early Adrenal Insufficiency Using Low-Dose Hydrocortisone in Very Preterm Infants: An Individual Patient Data Meta-Analysis Early (< 7 days) systemic postnatal corticosteroids for prevention of bronchopulmonary dysplasia in preterm infants Postnatal Corticosteroids To Prevent Bronchopulmonary Dysplasia
- Preparation of drugs in standard concentrations in the NICU
-
Preparation of drugs in standard concentrations in the NICU
Interesting. So the infusion pump gets the weight from the medical record automatically? If yes, then this removes another step of "manual" calculation, which surely contributes to less dosing errors. However, you still need to factor in the weight when the nurse connects the syringe to the pump and prepares to deliver the dose. If the drug solution is in mg/mL and the dose is given as a dosing rate (mg/kg*hour), then when dividing the dosing rate by the concentration you get mL/kg*hour, which is a weight-adjusted delivery rate. Is the weight factored in automatically by the pump, i.e., the pump calculates the final delivery rate in mL/hour?
-
Preparation of drugs in standard concentrations in the NICU
Dear colleagues, I was wondering what your practice is regarding preparation of drugs for intravenous administration in the NICU? Are your units employing an individualized approach, whereby each drug is prepared for each infant in the prescribed dose and thus administered, or a 'standard concentration' approach, whereby a drug solution is prepared in a fixed concentration and the volume to be administered is calculated from that concentration? In our unit we employ the latter approach, as can be observed in the attached figure. The doctor prescribes a weight-based dose, the pharmacist prepares a standard concentration solution in sufficient volume to account for dose and tubing, and the nurse calculates the volume that would provide the precise prescribed dose and sets the syringe pump to deliver that dose. I am interested to know of other units working this way, as well as other approaches. Please share for everyone's benefit!
-
Which resources do you use to check medication compatibility with breastfeeding?
Hi all, I am a neonatal and pediatric pharmacist from Israel, and I regularly provide consultations on drugs and breastfeeding in our NICU and obstetrics wards. I am delighted to see this discussion and the dyad-centered approach, which strives to take into account both mother and infant considerations and wishes to make an educated decision backed by careful risk management. This is without a doubt the right course of action! I will share the resources I am aware of and often use, and even though some have already been mentioned, perhaps this list may be useful as a convenient and accessible collection for your practices and everyday needs: Lactmed - An American, NIH-sponsored, freely available resource. Comprehensive and regularly updated. E-lactancia - A Spanish resource, available in English and Spanish, freely accessible. It should be noted that it also provides information on exposures other than pharmaceuticals (some food supplements, phytotherapy). MotherToBaby - An American resource powered by The Organization of Teratology Information Specialists (OTIS). They offer a vast collection of fact sheets for various exposures, including pharmaceuticals but many other exposures as well. While this is patient-oriented information, it may still be useful for professionals to observe the approach of OTIS to exposure with scarce evidence. Hale's Medications & Mothers' Milk - a well-known, long-standing American resource by Thomas Hale and Kaytlin Krutsch. Available digitally (to paying subscribers) and in print, this collection provides evidence summaries for many exposures (mostly pharmaceuticals, but not solely). One of its highlights is the pharmacokinetic data table that accompanies each monograph, which is very helpful for a quick observation, before diving into the evidence. It also contains some appendices that are worth checking out (drugs that may alter the taste of human milk and more). InfantRisk Center - This is the teratology information center in Texas Tech University that produces Hale's Medications & Mothers' Milk, and its online website and community offers additional resources and articles that may complement the book. I suggest checking out the forums, where patients and professionals may ask questions on exposures during breastfeeding and the InfantRisk team responds to them, and the breastfeeding page, which contains many articles on all kinds of exposures during breastfeeding. Academy of Breastfeeding Medicine - This American medical society publishes protocols on various topics related to breastfeeding, some include exposure to certain drug groups. Breastfeeding Medicine - This Journal, published by the Academy of Breastfeeding Medicine and Mary Ann Liebert, offers a myriad of peer-reviewed papers, protocols and publications on breastfeeding. Some material is open access, others require subscription access. It is possible to search the journal page for drug name/family and retrieve appropriate publications. This journal also features publications by Philip O. Anderson, who is also involved in Lactmed. His publications include summaries for various drug families in breastfeeding. Guidelines for anaesthesia and sedation for patients who are breastfeeding - This open-access British publication, which was originally published in 2020 and very recently revised and updated, includes a lot of information on many drugs used for analgesia and sedation in breastfeeding women. Be sure to check out the supporting information, the appendices offer much useful information (including more resources), basic terminology and more. SPS (Specialist Pharmacy Service) "Safety in Breastfeeding" topic - This NHS-supported service has a lot to offer about drugs and pharmacy skills in general, and there is a topic about "Safety in Breastfeeding" that serves as a collection of articles about pharmacotherapy during breastfeeding in general, as well as dedicated articles for certain drugs/drug groups (e.g., antihistamines, GLP-1 receptor agonists, contrast agents, oral anticoagulants, and more). Pharmacokinetics sections in drug labels and general drug databases - When all else fails, one may try to make an educated "guess" about the amount of drug that may pass into breastmilk according to PK parameters, e.g., low bioavailability of injectables, high molecular weight of biological products etc. This list is not complete, but provides a collection of trusted resources for healthcare professionals. I was also happy to see the paper @Annika shared, not only because I was a co-author on this paper, but also because I believe this review captures the 'modern' approach to drug exposure during breastfeeding and provides a holistic view of this topic that may allow NICU professionals to provide preterm infants with human milk that is SO important to them. So I do recommend reading it and applying our suggestions in your practices as you see fit. * A word on OpenEvidence - this is an excellent tool, the most trustworthy AI platform for medical information currently available. However, one should note that in questions about drugs, it often relies on information from FDA drug labels, which - as we know - often provide no safety information for exposure during pregnancy and/or breastfeeding, or even recommend against use of the drug in question. Therefore, always check the sources for OE answer, and if it relies on the label, do check in any of the other sources I listed above. Thank you for raising this topic, and I do hope this will contribute to better care for breastfeeding mothers and infants.
View in the app
A better way to browse. Learn more.
_0b6494.png)
_5c07e1.png)
_0d1bd3.png)
_75149d.png)